Research Outline
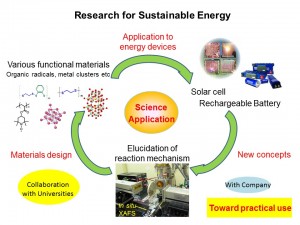
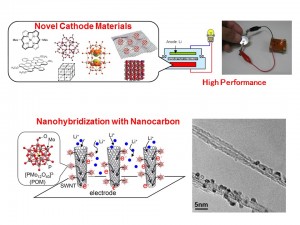
Recently, much attention has been focused on the creation of new energy systems, such as high-performance rechargeable batteries, as a solution to the global energy and environmental crises. Redox active molecular materials are good candidates for a high-performance cathode active material. To achieve both rapid charge/discharge and high power density, we have developed “molecular cluster batteries” using polynuclear metal complex clusters (molecular clusters), which exhibit multi-electron redox reactions, as a cathode active material for lithium batteries, and then achieved a larger capacity than that of the present Li ion battery using LiCoO2 as a cathode active material. Furthermore, to modify their battery performances, we also tried to nano-hybridize molecular clusters with nano-carbons such as single-walled carbon nanotubes, graphenes, and mesoporous carbons. The obtained nano-hybrid materials between molecular clusters and nano-carbons exhibited much larger capacities, compared with those of non-nanohybrid materials, due to the coexistence of redox reactions of molecular clusters and electrical double layer capacitors of nanocarbons enhanced by nanohybridization. These results indicated that development of redox active molecular materials and nanohybridization with nanocarbons are very important toward realization of next generation energy storage devices. From both viewpoints of applications and fundamental sciences, we are trying to investigate new materials and discover novel principles now.
Research Projects
- Development of novel rechargeable batteries using various molecular materials
- Modification of electrochemical energy storage performance by nanohybridization with nanocarbon
- Elucidation of battery reaction mechanism by synchrotron radiation